Research Question
How does the distance from the water inlet affect the pH level and the population of the microorganism present?
Hypothesis
As the water moves further from the water inlet, the pH will be higher and there will be more micro organisms because the water will have more exposure to plants which use the CO2 in the water, and because the water will be more still at locations further from the water inlet there will be more micro organisms.
Background Information
pH level
pH is the measurement of the acidity of water. Low pH indicates high acidity whereas a high pH indicates that the solution is alkaline.
Factors affecting pH
There are many ways that nature can affect the pH value, one of which is photosynthesis. photosynthesis is the process that plants use to convert the sunlight and carbon dioxide to glucose. The process requires carbon dioxide, which in water acts like carbonic acid (H2CO2). When carbon dioxide is used, the acidity of the water decreases, increasing the pH value. Thus the pH value of the water tends to be higher during the daytime. (PH)
Respiration and decomposition decrease the pH value. Respiration is the process that living thing breathe in the air and exchange the oxygen with carbon dioxide, consequently it decreases the pH value. Decomposition occurs when a living thing dies and the organism starts to decompose. The process releases carbon dioxide therefor decreasing the pH level of the surrounding water. (PH)
Lake and pH changes
As organisms and enzymes can only function optimally at certain pH levels, lakes have mechanisms to prevent changes in pH levels. This is called “buffering capacity” where small changes in pH are quickly modified by various chemical reactions. This is why the pH level of the water in the lake ranges around 6.5 to 8.5. (PH)
Microorganisms
Microorganisms gather up with mainly with 3 purposes which are: Light, pH level and resources
pH is the measurement of the acidity of water. Low pH indicates high acidity whereas a high pH indicates that the solution is alkaline.
Factors affecting pH
There are many ways that nature can affect the pH value, one of which is photosynthesis. photosynthesis is the process that plants use to convert the sunlight and carbon dioxide to glucose. The process requires carbon dioxide, which in water acts like carbonic acid (H2CO2). When carbon dioxide is used, the acidity of the water decreases, increasing the pH value. Thus the pH value of the water tends to be higher during the daytime. (PH)
Respiration and decomposition decrease the pH value. Respiration is the process that living thing breathe in the air and exchange the oxygen with carbon dioxide, consequently it decreases the pH value. Decomposition occurs when a living thing dies and the organism starts to decompose. The process releases carbon dioxide therefor decreasing the pH level of the surrounding water. (PH)
Lake and pH changes
As organisms and enzymes can only function optimally at certain pH levels, lakes have mechanisms to prevent changes in pH levels. This is called “buffering capacity” where small changes in pH are quickly modified by various chemical reactions. This is why the pH level of the water in the lake ranges around 6.5 to 8.5. (PH)
Microorganisms
Microorganisms gather up with mainly with 3 purposes which are: Light, pH level and resources
About the Nongbon Reservoir
The nongbon reservoir is a large man made lake connected to canals built in 1999. It carries 5,090,700 cubic meters of water and was built as a mechanism to relieve the flooding issue in Thailand. The lake is surrounded by plants, mainly grass and trees, and is used as a recreation centre due to its natural environment. The water rising and falling causes the shoreline to be damp and even slightly muddy without grass. Furthermore, there are fish carcasses (both old and fresh) on the grass, indicating the water level's potential of rising to that degree.
Apparatus and Methodology
Apparatus
Safety Equipment
- Gloves
- Lab coat
- Alcohol hand sanitiser
Other
- 5 Small containers
- 1 pH measurer (water)
- 1 pH measurer (soil)
- 500 ml Distilled water
- 1 Microscope with USB camera attached
- 5 pipettes
- 6 Beakers
- 1 haemocytometer (gridded slide)
- 1 slide cover
- 1 Computer
- Gloves
- Lab coat
- Alcohol hand sanitiser
Other
- 5 Small containers
- 1 pH measurer (water)
- 1 pH measurer (soil)
- 500 ml Distilled water
- 1 Microscope with USB camera attached
- 5 pipettes
- 6 Beakers
- 1 haemocytometer (gridded slide)
- 1 slide cover
- 1 Computer
Method
Collecting samples :
- Equip gloves and bring alcohol hand sanitiser in case of scrapes
- Starting from the water inlet, collect water samples every 100 metres. Use a measuring tape to measure 100 metres for precision
- Measure the soil pH and the water pH at the location and record it
- Using distilled water, rinse pH meter after every use.
pH after around 3 hours after collecting, in the lab :
- Label beakers 1-5 (one being 0m and 5 being 400m from the inlet) and pour the respective water sample into them
- Fill 6th beaker with distilled water
- Measure the pH of each water sample and record. Rinse pH meter after every use.
Finding microorganisms
- Label pipettes 1-5 and put them into the respective beakers
- Use different pipettes for different samples and drop the samples to the haemocytometer
- Use the microscope and find the focus point and the grid.
- When found, project image on screen and save the image to count how many organism are in the water samples
- Clean the haemocytometer with distilled water and dry with paper after use and repeat the step above for the remaining 4 samples
Variables
Independent
Distance of water sample from water inlet
Dependent
pH level of the water and the population of microorganisms
Controlled
Magnification of microscope and Number of squares counted
Distance of water sample from water inlet
Dependent
pH level of the water and the population of microorganisms
Controlled
Magnification of microscope and Number of squares counted
Raw Data
Data table
Distance from Inlet (m) |
Water pH at reservoir |
Soil pH |
Water pH 3 hours later |
o |
6.72 |
7.4 |
6.39 |
100 |
7.46 |
7.6 |
6.39 |
200 |
7.53 |
7.5 |
6.47 |
300 |
7.61 |
7.5 |
6.72 |
400 |
7.49 |
7.6 |
7.06 |
Further observations :
|
|
Processed Data
|
|
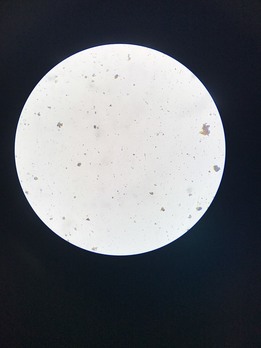
Images displayed on microscope projected onto the computer screen
|
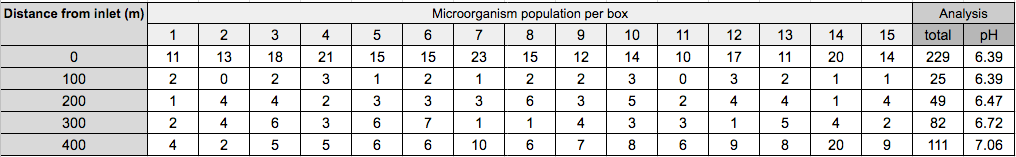
Table 2 : Microorganisms per box of each water sample
Table and graphical representation of correlation between distance from inlet and population of microorganisms
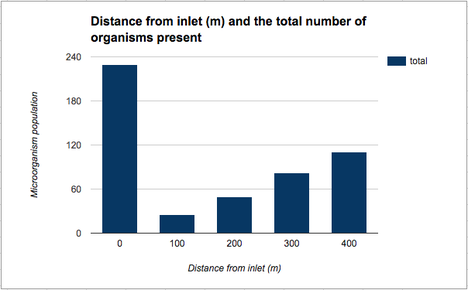
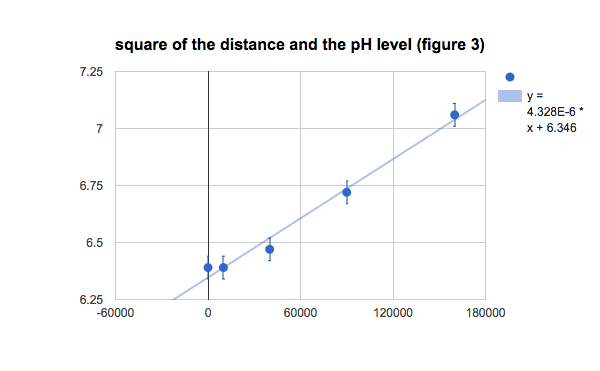
Figure 2 :Graph showing correlation between distance and population
Analysis
The pH level of the water is lower when the the water was contained in a container for a couple of hours. This is because of the respiration of the microorganisms present in the water samples.
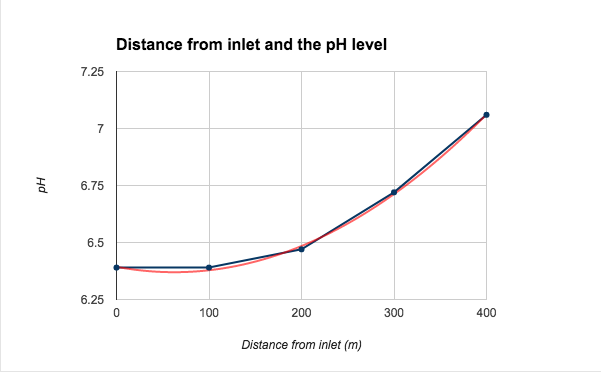
Based on the graph, distance from inlet and the pH level of the water, the pH level increases as the water moves further from the watergate. The rate of increase (slope) is not constant, this nonlinear trend shows an exponential relationship of the water pH level according to the distance away from the water inlet. The figure 3 also point out this relationship since the square of the distance is proportional to the water pH level. Although there are some differences in the values, making them not exact, when including the error bars of 0.005 (from the pH meter), all the data points hit the line of best fit. This relationship indicates that acidity level of the water decreases as the distance from the water inlet increases.
The population of the microorganism does not show clear correlation to the distance nor the water pH. The pH graph shows an overarching trend of increase in pH as the water moves further from the source. The population of the micro organism, however, does not. This is mainly due to the high population in the water sample at the point of O meters. The other 4 samples show a gradual increase as the distance from the water inlet increases. The environmental factors of these four water samples are similar ; being that the number of fish carcasses are fairly similar (0 or 1) whereas in water sample 1 there were 5 or more dead fishes.
The garbage stench from the proximity of the water source may be due to the canal water seeping in. As there were signs of human residence, one can assume the disposal of waste products into the water canal. The water seeping in may carry micro organisms with it, affecting the population of the micro organisms.
Conclusion
Population of Microorganism
|
Based on the background information regarding microorganisms, they concentrate around food sources. As the further observation section mentions, there were two dead fishes floating in the water near the location where sample 1 was collected, and there were at least 3 dead fishes in the water gate. Based on these observations, one can conclude that the high population of the microorganisms is not related to the distance from the water inlet but the surrounding environment, mainly fish carcasses. The stench of the canal water seeping in through the soil may also be a factor in increasing the population of the microbes.
In conclusion : the distance from the water inlet does not affect the population density of the microorganisms present in the water. |
pH level
|
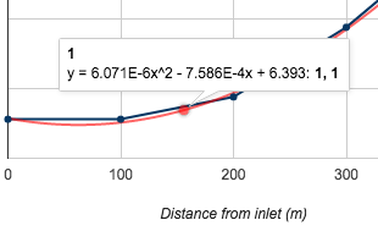
The pH level, unlike the population of the microorganisms present in the water samples, shows a direct correlation to the distance from the water inlet. As the distance from the water inlet increases, the pH level of the water sample increases in a exponential manner of
y = 6.071E-6x^2 - 7.586E - 4x + 6.393 This could be due to the exposure to more plants and autotrophic microorganisms causes the carbon dioxide dissolved in the water to be used during the process of photosynthesis. |
Thus, our hypothesis was incorrect.
The answer to our research question is :
"The increase in distance decreases the pH and does not have direct correlation to the population density of the microorganisms present in the water body."
The answer to our research question is :
"The increase in distance decreases the pH and does not have direct correlation to the population density of the microorganisms present in the water body."